Atoms and molecules
Science Wednesday
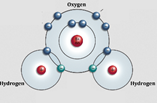
1. Atoms are made of tiny particles that are constantly in motion.
2. These particles have electrical charges – a little zing! – and attract particles from other atoms & molecules.
3. FOR EXAMPLE: Magnets are made of elements that have a strong positive charge on one side and a strong negative charge on another.
4. DID YOU KNOW: Water molecules are a tiny bit magnetic too. Notice how water molecules stick to each other and to anything you dissolve in water.
|
Challenge 1: Try out a magnet:
(a.) Use any of the magnets on the table to connect to other magnets or to pick up pieces of metal
|
|
Challenge 2: Make a cup of tea:
A. Dissolve tea in a cup of warm water
B. THINK LIKE A SCIENTIST: Imagine that each water molecule and each water droplet is like a magnet, attracting the particles of tea.
|
|
Challenge 3: Over-fill a container with water
(a.) Use a dropper to fill up a container with water.
(b.) Keep adding more water to see if you can fill the container higher than the edges without spilling.
(c.) THINK LIKE A SCIENTIST: Why does this work?
|
|
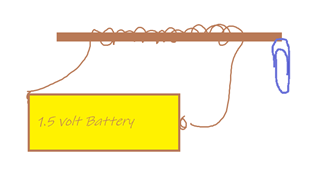
Challenge 4: Make an electromagnet:
(a.) Wrap wire around a straight piece of metal (bolt or nail).
(b.) BRIEFLY attach both ends of the wire to a battery
(c.) Test to see if the electromagnet will pick up a paperclip
(d.) Quickly disconnect the electromagnet before it gets too hot.
(e.) Use cardboard to design a frame for the electromagnet.
(f.) THINK LIKE AN INVENTOR: How could you use an electromagnet to make something useful?
|
|
Challenge 5: Un-make a cup of tea:
(a.) Can you use some type of filter to turn the cup of tea back into clear water?
(b.) THINK LIKE AN INVENTOR: How can you use filters to make something useful?
|